# Major Recall Alert: Medline Convenience Kits Containing Namic RA Syringes Recalled Due to Potential Safety Risk
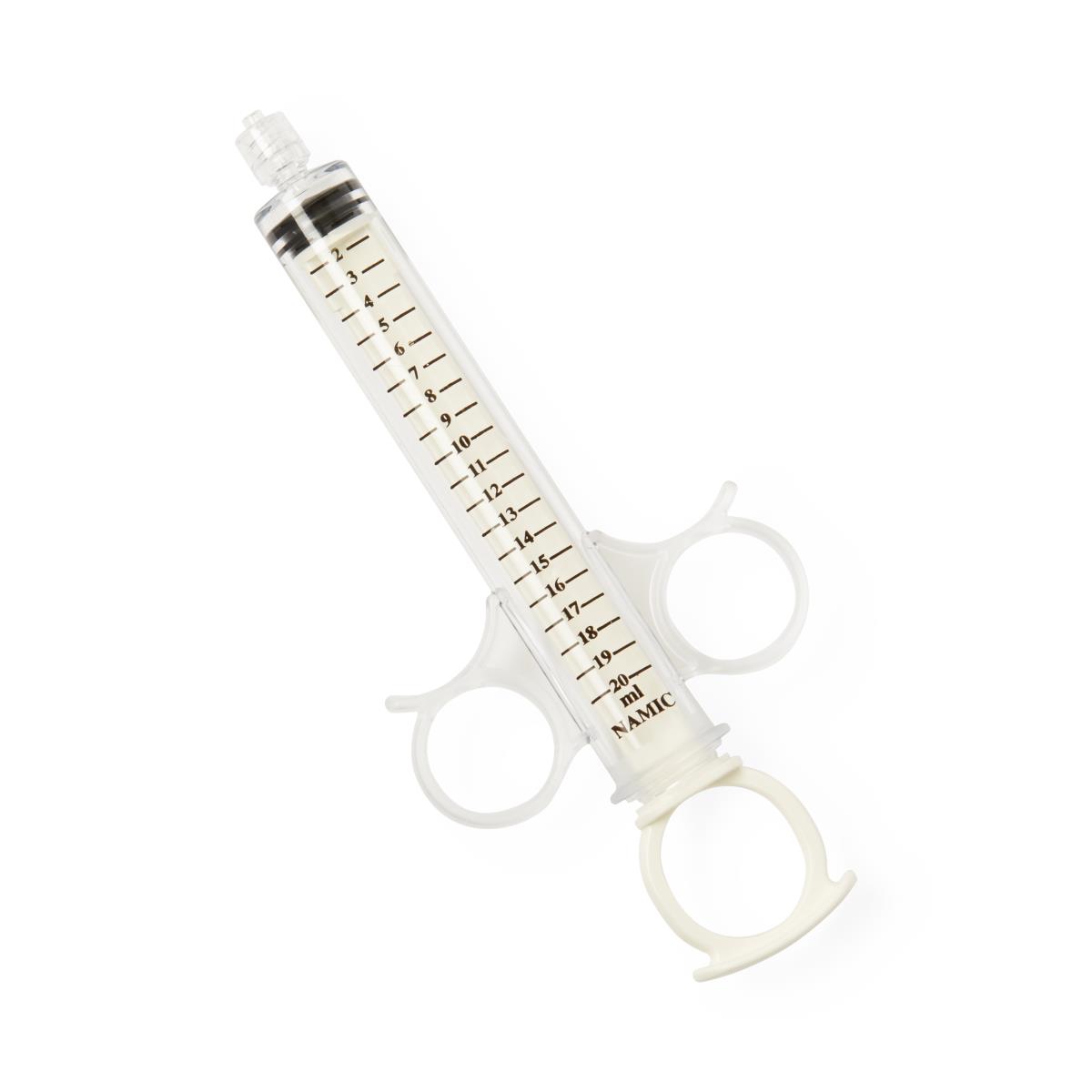
Medline Industries has issued an urgent recall for various convenience kits containing **Namic Angiographic Rotating Adaptor (RA) Control Syringes (Namic RA Syringes)**. This recall has been initiated due to a potential defect in the syringe's rotating adaptor, which can unwind during use. If this occurs, it can lead to significant procedural delays or, in severe cases, life-threatening consequences, including death. Affected groups have been strongly advised to stop using these syringes immediately and dispose of them properly to ensure patient safety.
Keep reading for important details on the recall, why it matters, and how you can take action to protect yourself and your loved ones.
---
## Why This Recall Is Important
Safety recalls like the one issued for the Namic RA Syringes underline the critical importance of avoiding any product that could compromise patient health. The Medline recall highlights the following risks:
- **Functional Defect**: The rotating adaptor of the syringe may become unwound, disrupting medical procedures.
- **Health Risks**: The defect can cause procedural delays that may escalate into severe medical emergencies or, in extreme cases, death.
- **Broad Impact**: The recalled kits are used in important angiographic and tumescent procedures in various healthcare settings.
Healthcare professionals and patients alike rely on these kits, making it essential for all users to be aware of the potential risks and recall instructions.
---
## Details of the Recall
This urgent recall impacts several product kits, including:
- **LHK Kit**
- **CA Kit**
- **Southlake Regional Kit**
- **Namic Convenience Kit**
- **Namic Custom Angiographic Kit**
- **Tumescent Syringe Kit**
All these kits contain Namic Angiographic Rotating Adaptor (RA) Control Syringes, which pose the safety issue.
### Official Recall Information:
- **Date Announced**: October 2023
- **Reason **: Risk of the rotating adaptor unwinding during use.
- **Advice to Consumers**: Immediately stop using Namic RA syringes and discard them from the affected kits to prevent potential complications.
For the official government notice on this recall, visit the [Health Canada Recall Notice](https://recalls-rappels.canada.ca/en/alert-recall/medline-convenience-kits-containing-namic-ra-syringes).
---
## What You Should Do
If you’ve purchased or used any of the kits listed, follow these steps to ensure safety:
1. **Stop Using the Affected Product**
Identify if your syringe kit contains Namic RA Syringes. Immediately stop using it if affected.
2. **Properly Discard the Syringe**
Safely dispose of the syringes and consult your original supplier for further instructions on replacing affected components.
3. **Contact the Manufacturer or Health Provider**
Reach out to Medline Industries or your healthcare provider for additional guidance.
4. **Share This Information**
Inform colleagues, healthcare facilities, or others who might be using these kits.
---
## Stay Safe – Get Instant Recall Alerts
With recalls like this potentially affecting lives, staying informed is critical. Don’t wait to take action—track recalls as soon as they’re announced by downloading our **MyRecalls App**. With real-time notifications, you’ll never miss updates on products that could impact your health or safety.
**[Download the MyRecalls App Now](#)** and share this lifesaving tool with those who need it most.
Stay safe and recall aware!