# Major Recall Alert: Soft-Vu and Accu-Vu 4 fr Angiographic Catheters Recalled Due to Manufacturing Defect
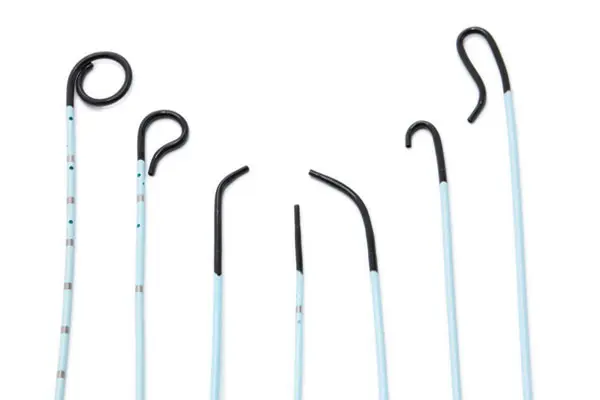
AngioDynamics, Inc. has issued a **voluntary recall and field safety corrective action** for specific **Soft-Vu and Accu-Vu 4 fr Angiographic Catheters**. This recall stems from a manufacturing defect involving the molded hub of certain catheter models and lots. The defect may prevent appropriately sized guidewires from passing through the catheter's inner diameter (ID), potentially impacting device usability.
While complaints associated with this issue have been reported, **no injuries or adverse events have been documented in Canada**, and no patient harm has been reported to date. Below is a summary of the recall and what it means for healthcare providers and patients.
---
## Why This Recall is Important
Angiographic catheters, such as the **Soft-Vu and Accu-Vu**, are critical medical devices used for diagnostic and therapeutic procedures. Safe and reliable performance is essential to ensure the success of these procedures. **A manufacturing defect in the molded hub** of certain Soft-Vu and Accu-Vu 4 fr catheters could result in serious consequences, including:
- **Inability to complete medical procedures:** The guidewire may not pass through the catheter properly, potentially delaying treatment.
- **Increased risk of complications:** If the device fails, medical professionals may need to choose alternative methods or devices, increasing the risk of procedure-related complications.
AngioDynamics has initiated this recall proactively to prevent any such occurrences, despite no known injuries linked to this issue in Canada.
---
## Details of the Recall
Here’s everything you need to know about the affected products:
- **Recalled Products:**
- Accu-Vu Sizing Catheter - Pigtail Flush
- Soft-Vu Angiographic Catheters: Shepherd Hook (Braided & Non-Braided)
- Soft-Vu Angiographic Catheters - Kumpe
- Soft-Vu Angiographic Catheters - Omni Flush
- Soft-Vu Angiographic Catheters: Berentstein (Braided & Non-Braided)
- Soft-Vu Angiographic Flush Catheters: Pigtail
- Soft-Vu Angiographic Selective Catheters: Cobra (Braided & Non-Braided)
- **Reason for Recall:**
A **non-conformance of the molded hub** in specific catheter models and lots prevents the passage of an appropriately sized guidewire through the inner diameter (ID) of the catheter hub.
- **Complaints Reported:**
Multiple complaints related to the issue have been confirmed. However, **no associated cases or adverse events have been reported in Canada**.
For official details, visit the [Canadian Government Recall Notice](https://recalls-rappels.canada.ca/en/alert-recall/soft-vu-and-accu-vu-4-fr-angiographic-catheters).
---
## What You Should Do
If you are a healthcare provider or end-user in possession of the affected devices, **AngioDynamics recommends the following actions**:
1. **Identify affected products**: Check your inventory for the specific Soft-Vu and Accu-Vu 4 fr catheter models and lots subject to the recall.
2. **Discontinue use immediately**: Stop using the identified products to avoid potential procedural or patient risks.
3. **Contact AngioDynamics**: Reach out to the manufacturer for further instructions on product returns or replacements.
4. **Report complaints or adverse events**: Healthcare professionals are encouraged to report any device malfunctions or adverse outcomes to local health authorities or directly to AngioDynamics.
---
## Stay Safe – Get Instant Recall Alerts
For healthcare professionals and patients, staying informed about medical device recalls is critical. Avoid potential risks by ensuring you’re up to date on the latest safety news.
**Download our free mobile app now** to receive **real-time alerts** about recalls in Canada. Stay proactive about your health and safety, whether it’s medical devices, food, or consumer products.
[CLICK HERE TO DOWNLOAD THE APP](#)
Stay informed, stay safe!
---
For more information on this recall, refer to the official notice on the [Canadian Government Recall Website](https://recalls-rappels.canada.ca/en/alert-recall/soft-vu-and-accu-vu-4-fr-angiographic-catheters).
*Image Source:
*